
This article summarises ideas and a model to explain ME/CFS published in the Journal of Primary Health Care (2019) by Angus Mackay. Mackay writes from the perspective of being a long-term sufferer and researcher of ME/CFS. Mackay explains the onset, symptomology and flare ups experienced by those with ME/CFS, and builds on the model proposed by Mackay and Tate (2018) in a previous article.
After becoming unwell with glandular fever (1995), which led to ME/CFS, Mackay tried to make sense of his continual flare ups and recovery cycles. Mackay had previously led an active life including time served as a British army officer, biology teacher and rugby coach. Over many years he carefully logged his symptoms and their severity, and what was triggering them to “flare up”. Mackay noticed a perceived intensity of brain and spine inflammation every time he experienced a symptom flare up, which would slowly dissipate as symptoms decreased.
He later dedicated time as a researcher in Professor Warren Tate’s laboratory, at the University of Otago, from 2011-18, to see if he could determine the root cause of ME/CFS. Due to the arduous nature of lab work, Mackay turned his attention to online literature exploring alternative theories to the chronic viral hypothesis that they were focusing their attention upon. Mackay searched the literature according to his growing conviction that ME/CFS was a neurological disease with neuro-inflammation of the brain being at its core. Mackay highlights even in its name “encephalomyelitis” meaning “inflammation of the brain and spinal cord” indicated many before him had thought the same way. But where was the evidence? Blood-biomarker studies appeared inconsistent and unreliable, to Mackay, while brain-imaging studies lacked the sensitivity to detect what Mackay perceived as a low-level, but chronic inflammation in the brains of ME/CFS patients.
Breakthrough study
Mackay initially focussed on a Japanese research group who showed, for the first time, clear evidence of inflammation in the brains of people with ME/CFS, using position emission tomography / magnetic resonance imaging (PET/MRI). The group published a paper (2014) showing activated glial cells (immune cells of the brain) in people with ME/CFS. Activation of the glial cells characterisers an inflammatory response and appeared to be in proportion to symptom severity e.g. levels of patient fatigue. Nearly all inflammation pertained toward the limbic system (mood-centre) in the brain of ME/CFS patients (see diagram).
Further (more recent) supporting evidence
Younger’s research (2018) also supports this finding of inflammation within the brain, highlighting the limbic system, using a different kind of brain scanning technique. Their technology detected lactate and small temperature increases in the brains of ME/CFS patients, both indicative of chronic inflammation. During this study healthy controls were found to have NO lactate or temperature changes within their brains. A recent PET/MRI study (2018) has also detected inflammation in the brains (also highlighting the limbic system) of people with Fibromyalgia, a very closely related disease to ME/CFS.
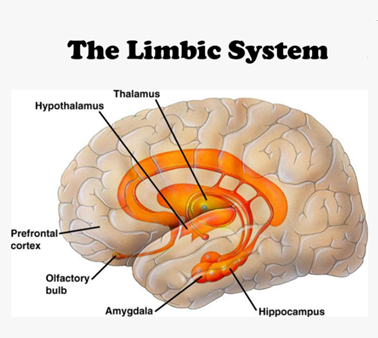

If the limbic system, and hypothalamus are inflamed and dysfunctional, this causes the ANS to be disrupted also. This could explain the majority (of the wide range) of symptoms experienced by those with ME/CFS.
Flare ups explained
Mackay noticed (and as recorded in the literature for ME/CFS patients, in general) that his symptoms waxed and waned according to a range of potential stressors, which included physical, mental, emotional, financial stress, infections and vaccinations, and environmental stress, including sleep deprivation or chemical toxins exposure. His “eureka” moment came when he found a piece of research that indicated that this range of stressors, following different physiological pathways all targeted the hypothalamus – suggesting that the hypothalamus acts as a kind of “stress response centre”, which processed these incoming “stress signals” from a variety of sources, before responding either hormonally (via the blood) or by electrical impulses (via the ANS).
Triggers Explained
Mackay intuitively realized that the triggers for ME/CFS like the stressors, described above, could also target the hypothalamus and set the disease off, if the stressors were severe enough and the person was ME/CFS susceptible. And the triggers which are known to trigger ME/CFS are indeed all of the “severe” kind, be they viral e.g. glandular fever, chemical toxins or extreme trauma.
(It is interesting to note that Covid19 appears to be triggering ME/CFS in a small percentage of patients… no surprise to Mackay, who classes Covid19 as just another on the list of “severe stressors” that can shock the hypothalamus into dysfunction).
From his research findings Mackay posed the following questions within his article:
- Might an inflamed (dysfunctional) hypothalamus help to explain why those with ME/CFS have a lower tolerance to stress/stressors?
- When a certain threshold for incoming stress signals is exceeded; could this trigger a flare up (relapse) originating in the hypothalamus and spreading out to specific targets within the brain, spine and ANS?
- Could the hypothalamus be a key vulnerable site in those predisposed to ME/CFS?
- May this then be the site of a genetic malfunction fundamental to the disease becoming manifested?
- Might this then trigger a neuro-inflammatory response within the brain of a person with ME/CFS?
- And might this inflammation then be self-perpetuated indefinitely by a now dysfunctional (inflamed) hypothalamus, no longer able to tolerate even the smallest amount of incoming “stress signals”, from a variety of sources (stressors)?
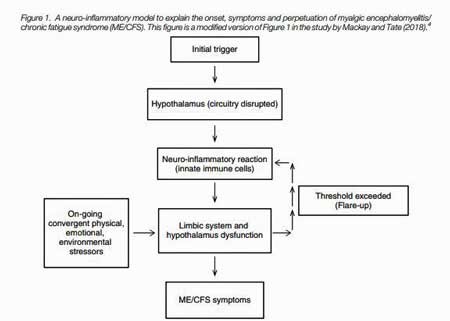
Although Mackay notes that this model is largely theoretical at this stage, it is hoped that it’s coherent nature could be used as a framework for future scientific research.
To view the full article click on the following link https://www.researchgate.net/publication/337624219_A_neuro-inflammatory_model_can_explain_the_onset_symptoms_and_flare-ups_of_myalgic_encephalomyelitischronic_fatigue_syndrome
Reference
Mackay, Angus. (2019). A neuro-inflammatory model can explain the onset, symptoms and flare-ups of myalgic encephalomyelitis/chronic fatigue syndrome. Journal of Primary Health Care. 11. 10.1071/HC19041.
Article summary written by Charlotte
